|
12/16/2023 0 Comments Element table charges 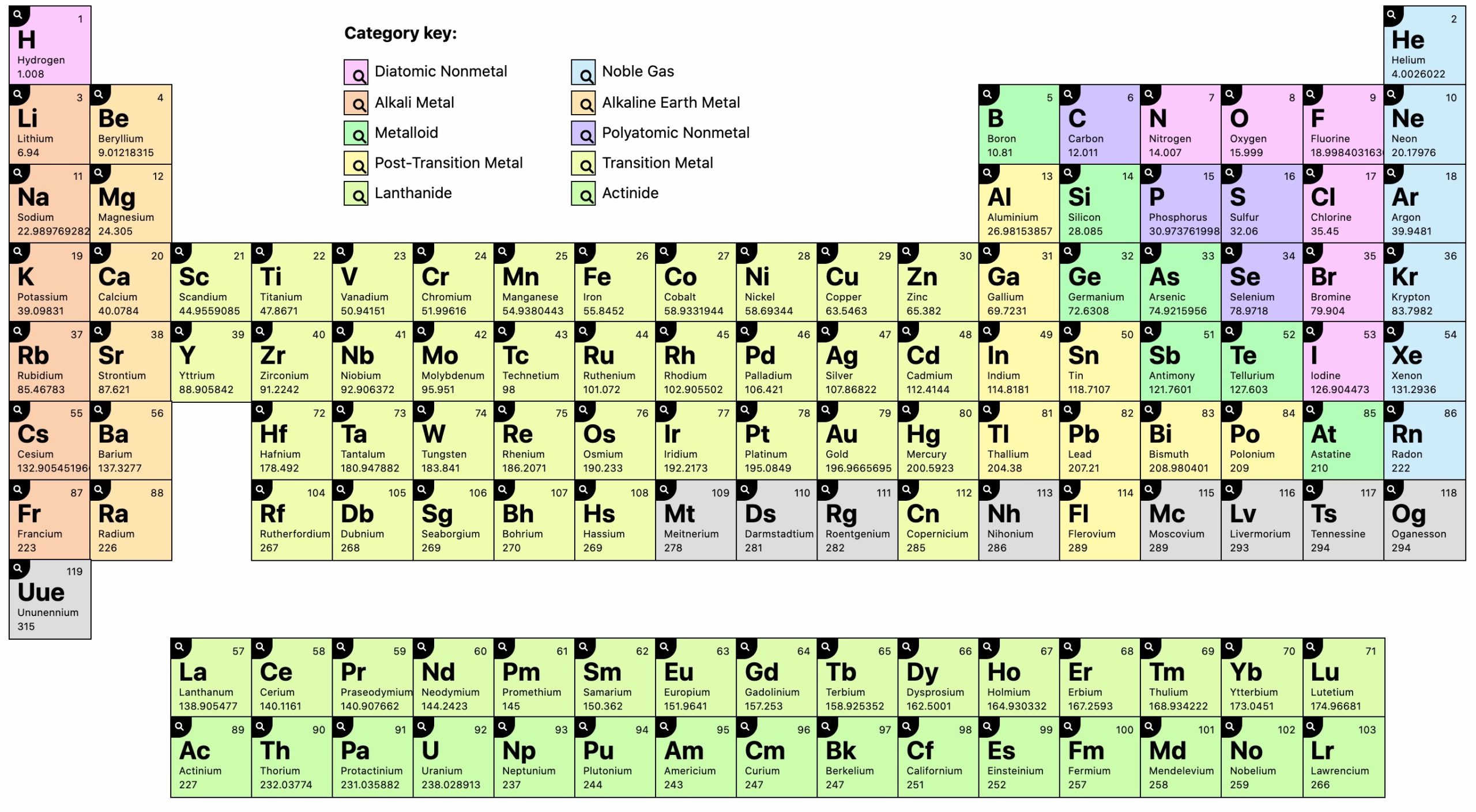 
Lund (Anoka-Ramsey Community College), Melissa Alviar-Agnew, and Henry Agnew. This page is shared under a CK-12 license and was authored, remixed, and/or curated by Lance S. 
The color periodic table with charges also have the most valence charges for the elements.\): The Modern and Common System of Cation Names Element It is also for the teacher aided needs for teaching their students and they also forget some points which are important to teach to the students but forget sometimes so they can write their points and can easily remember those points at the time of teaching as they can carry that periodic table with charges easily with them and can also paste it on the walls of the classroom.įind Our Periodic Table Valency Of Elements. They can write those important points and can carry along with them and can also easily remember. Because of the effects of shielding and the different. Except for hydrogen, Zeff is always less than Z, and Zeff increases from left to right as you go across a row. As will be discussed in more detail later in this chapter, atoms (and molecules) typically acquire charge by gaining or losing electrons. In this periodic table with charges templates we are providing you with different colors and combination which will be very useful for your use and also available with different size so that you can choose according to your need and also it has some spaces toward bottom right side so that you can add some important notes if you have any for those students who have the habit of forgetting some little important points because of the pressure of the other work or due to their busy schedule so it will help both of them. Figure 2.5.1: Relationship between the Effective Nuclear Charge Zeff and the Atomic Number Z for the Outer Electrons of the Elements of the First Three Rows of the Periodic Table. Nuclear charge is the positive charge present in the nucleus of an atom, which is equal to the number of protons in the nucleus, i.e., the atomic number. The charge of an atom is defined as follows: Atomic charge number of protons number of electrons (1.8.1) (1.8.1) Atomic charge number of protons number of electrons. On the periodic table meats are founded on the right and all the non-metals are founded on the right side which are negative but for that you also need to have information regarding the elements of the ionic charge. Periodic Table with charges means to find out the ionic charges of the element in the periodic table. That is why the d-block of the periodic table figure above has been labeled variable. If you can download the other Article of Periodic Table with Name of Element And Periodic Table Valency Of Oxygen. Most transition metals, however, can form cations of various charges. You can use it whenever you need anything regarding any issues of chemistry. At the bottom of the table there is two row block of the elements which have actinides and lanthanides. It will give you all the information regarding the electronegativity, elements symbols or elements square its valence number and its electron configuration. Because this element is located in Group 17, or 7A, on the periodic table, it will ionize to form an anion with a 1 charge. 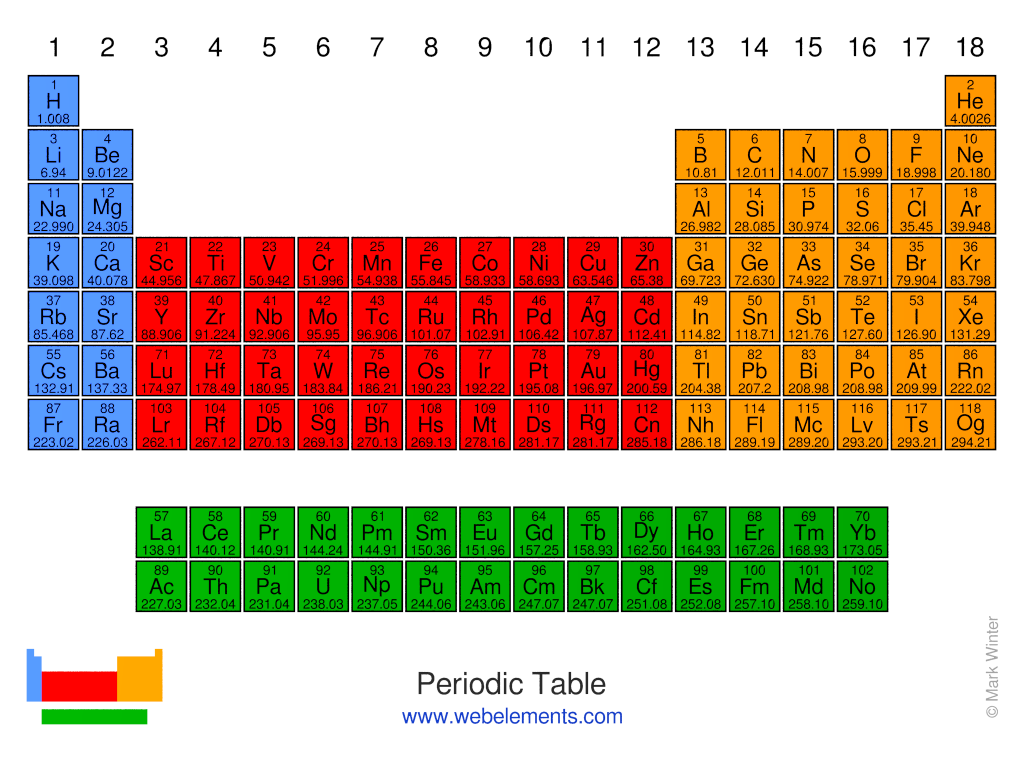
The number of protons, neutrons and electrons will help you to read the elements of the periodic table where all the information regarding the number and symbols are also displayed in the periodic table. The remaining columns each have an associated positive or negative numerical value that indicates the charge that results when elements in that column are ionized. Firstly we will discuss something about the periodic table what is it and then it uses Periodic table is known as the arrangement of all the chemical elements in tabular way with all the information likewise atomic number, recurring chemical properties and its electron charges which will show periodic trends. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
